How to Compare qpcr and pcr for Your Research Needs?
In the world of molecular biology, comparing qPCR and PCR is crucial for research advancements. As Dr. Emily Carter, a leading expert in genetic analysis, once said, "Choosing between qPCR and PCR can shape the future of your research." This statement resonates deeply in the scientific community where the differentiation between these techniques can determine the success of a project.
Real-time quantitative PCR (qPCR) offers precise quantification of nucleic acids. In contrast, traditional PCR focuses primarily on amplification. Researchers must weigh the benefits of sensitivity and specificity. Understanding these methods is not always straightforward. Numerous factors must be considered, such as the target sequence, sample quality, and desired outcomes.
With the complexity of each method, mistakes can happen. Misinterpretation of results can lead to flawed conclusions. This highlights the importance of careful analysis. In a world where every data point matters, reflecting on the choice between qPCR and PCR is essential for delivering impactful research outcomes.

Understanding the Fundamental Differences Between qPCR and PCR
qPCR and PCR serve different purposes in research. PCR amplifies DNA, allowing you to produce millions of copies. It’s a simple technique, widely used for cloning and gene expression analysis. On the other hand, qPCR quantifies DNA in real-time. This method provides data on the initial amount of DNA present in your sample. Understanding these differences is crucial for choosing the right method for your experiments.
When selecting between qPCR and PCR, consider your specific research goals. If you need to measure gene expression levels, qPCR is preferable. For basic amplification tasks, traditional PCR suffices. Each technique has strengths and weaknesses that may impact your results. Be mindful that qPCR can be more costly and requires specialized equipment.
Tips: Always validate your results. Run controls to ensure accuracy. Check for non-specific amplification in PCR. Don’t skip optimization steps; they can drastically alter your outcomes. It's essential to critically reflect on the protocols used and adapt them as necessary. Sometimes, revisiting your approach opens new avenues for discovery.
Analyzing Sensitivity and Specificity in qPCR Versus PCR
Quantitative PCR (qPCR) and conventional PCR serve distinct roles in molecular biology. qPCR is known for its sensitivity, allowing for the detection of low copy numbers. It achieves this by amplifying DNA while also measuring the amount in real-time. This is especially useful in applications like gene expression analysis. On the other hand, traditional PCR is less sensitive and primarily focuses on generating enough DNA for downstream applications. While qPCR provides quantitative data, PCR only gives a qualitative result.
Specificity is another critical factor to consider. qPCR is designed to reduce non-specific amplification, yet it can still yield false positives if the primers are not carefully selected. In contrast, traditional PCR often struggles with non-specific products. Researchers have faced challenges when their PCR reactions produce undesired bands on gels. This can lead to misinterpretation of results. It's essential to optimize conditions to minimize non-specific interactions, regardless of the method chosen.
Balancing sensitivity and specificity is crucial for any experiment. While qPCR may seem like the more favorable option, its complexity demands robust protocol development. Simple mistakes can lead to misleading results. Likewise, standard PCR can serve basic needs, though it may lack the precision required for certain applications. Each method has its pros and cons, which can only be fully realized through thoughtful experimentation.
How to Compare qPCR and PCR for Your Research Needs?
| Parameter | qPCR | PCR |
|---|---|---|
| Sensitivity | High (10-100 copies) | Moderate (100-1,000 copies) |
| Specificity | Very High | Moderate (Dependent on primers) |
| Quantification Capability | Yes | No |
| Time Efficiency | Shorter (2-3 hours) | Longer (3-5 hours) |
| Cost | Higher | Lower |
| Equipment Requirement | Requires qPCR machine | Standard thermal cycler |
Evaluating Quantification Accuracy in qPCR for Research Applications
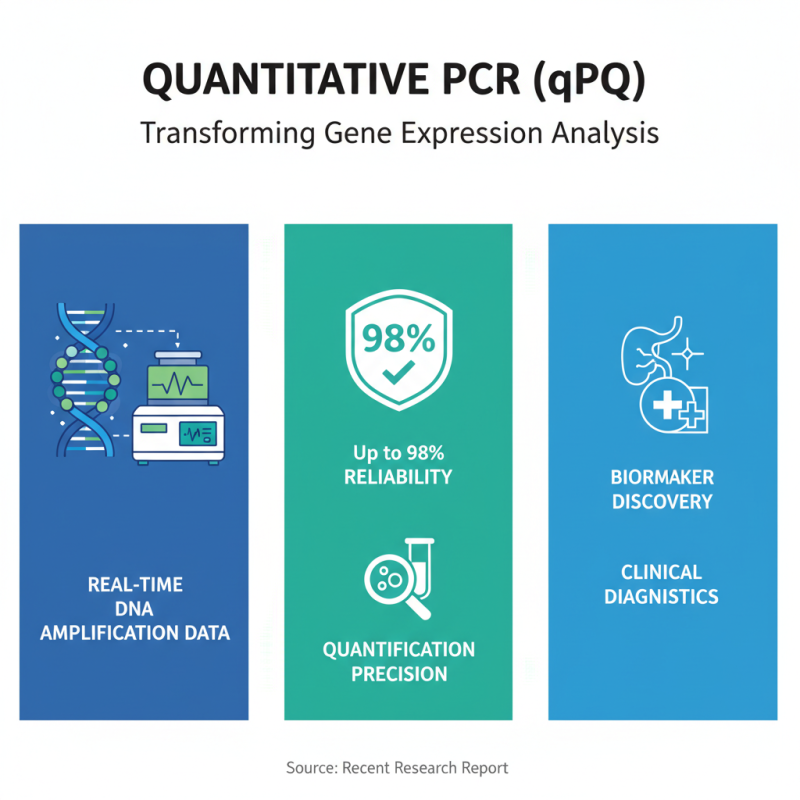
Quantitative PCR (qPCR) has transformed how researchers assess gene expression. It offers real-time data on DNA amplification, providing valuable quantification accuracy. A recent report indicates that qPCR can achieve a reliability of up to 98% for quantifying target sequences. This precision is crucial for applications like biomarker discovery and clinical diagnostics.
However, qPCR isn't without challenges. Variability in reagents can influence results. A study showed that 20% of laboratories reported inconsistent RNA extraction protocols, impacting quantification outcomes. This inconsistency highlights the need for careful method validation. Researchers must be vigilant about their experimental conditions to ensure reliable data. Adjustments in primer design can also affect the amplification efficiency.
In contrast, traditional PCR remains relevant for certain applications. It does not provide quantitative data like qPCR but is often simpler and less resource-intensive. While qPCR provides quantitative results, it may not be necessary for all research fields. The choice between qPCR and PCR hinges on the specific research question and the required accuracy. Therefore, continuous evaluation of both techniques is essential for optimal research outcomes.
Cost-Benefit Analysis: qPCR vs. Traditional PCR Methods
When comparing qPCR and traditional PCR methods, a cost-benefit analysis is essential. Recent studies reveal that qPCR, although initially more expensive, can offer significant savings in the long run. The initial cost of qPCR reagents may be 30-50% higher. However, due to its increased sensitivity and speed, researchers often achieve results faster.
qPCR allows for real-time monitoring. This feature minimizes wastage, as less sample material is needed. Traditional PCR may require multiple cycles, leading to higher consumable costs. A survey showed that over 60% of laboratories reported costs associated with reagents and time are significantly lower with qPCR.
However, qPCR systems have a higher upfront investment. This can deter smaller labs with tight budgets. Some researchers also note that not every experiment requires the precision of qPCR. Relying solely on qPCR for every assay may lead to a skewed allocation of resources. Balancing project requirements with budget constraints is key for effective research management.
Optimizing Protocols for Enhanced Performance in qPCR and PCR
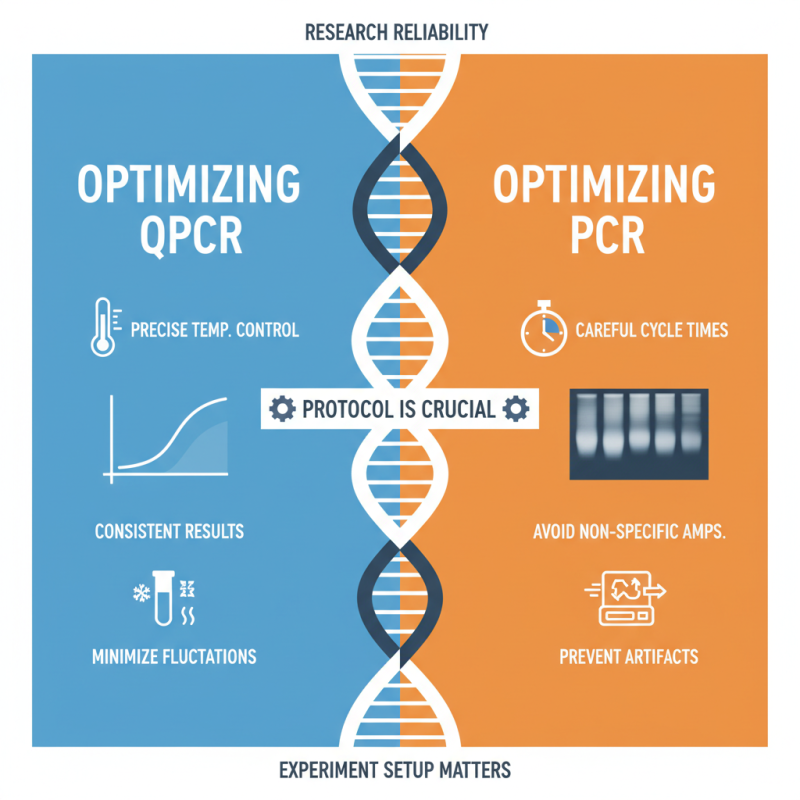
Optimizing qPCR and PCR protocols is crucial for researchers. Each technique has its strengths, but performance relies heavily on how you set up the experiments. For qPCR, maintaining precise temperature control is essential. A minor fluctuation can lead to inconsistent results. Meanwhile, traditional PCR often demands careful attention to cycle times to avoid non-specific amplification.
Consider this tip: always run a few test reactions to gauge your conditions. This can reveal hidden issues with primer binding or enzyme activity. It helps refine your approach without investing too much time upfront. Similarly, for qPCR, utilizing appropriate reference genes can significantly enhance your data’s reliability.
Pay attention to sample quality as well. Contaminants in your DNA can affect both methodologies. Experimenting with different extraction protocols might help you find the best fit for your samples. Remember to index your data and protocols well; it saves time during analysis. Finding that balance between precision and practicality is often where the real learning happens.
Related Posts
-

Why is qPCR Important for Modern Biological Research?
-
Why is DNA PCR Important for Genetic Testing and Research?
-
How to Effectively Understand the PCR Steps in Molecular Biology?
-

What is qPCR Polymerase and How Does it Work in Genetic Analysis?
-

2026 Best Q PCR Techniques for Enhanced Performance and Accuracy?
-

Why is PCR and RT PCR Important in Modern Diagnostics?